VOLUNTARY CERTIFICATION
Voluntary Certification of products is carried out in the sphere unregulated by law, when the products are not subject to the mandatory European directives.
The process of voluntary certification is relatively simple. It consists of the following steps:
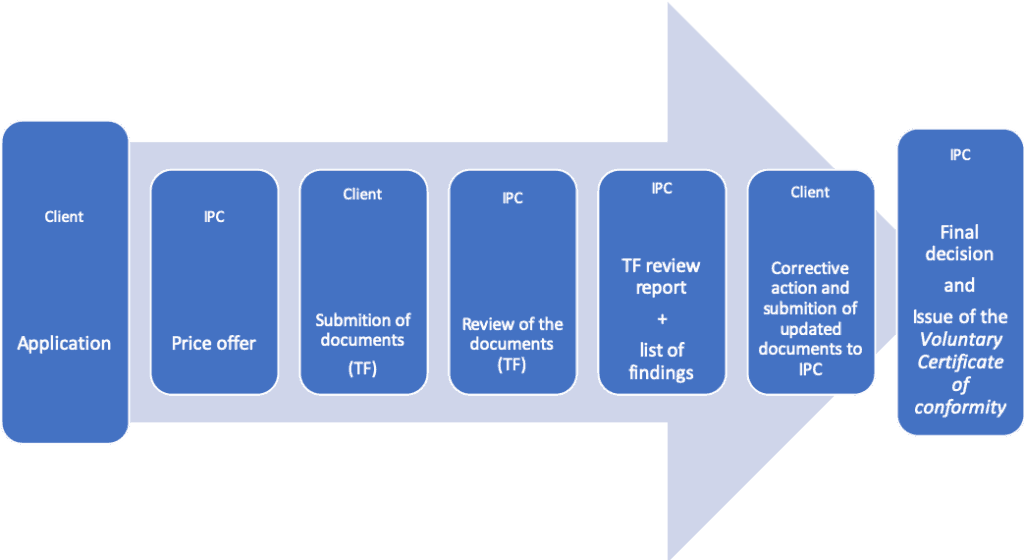
In case of positive results from the Documentation Review (TF), a certificate of voluntary certification and conformity to European standards is issued.
The certificate is valid for 3 years unless there are any changes in the technical documentation or changes in legislation, directives.
Why is the Voluntary Certification valuable to you?
- You will get an independent and impartial opinion
- confirmation of own assessment and avoiding later losses due to complaints or non-compliance with legal requirements
- getting a better position on the competitive domestic and foreign market, and thus facilitating sales
- gaining an advantage in tenders or public procurement
Product Certifications: For European market, CE mark certification services are provided as per EU Regulations and EC Directives;
- 2014/35/EU – Low Voltage Directive (LVD)
- 2014/30/EU – Electro Magnetic Compatibility
- 2006/42/EC – Machinery Directive (MD)
- 2014/68/EU – Pressure Equipment Directive (PED)
- 2014/34/EU – Equipment and protective systems intended for use in potentially explosive atmospheres (ATEX)
- 2011/65/EU – Restriction of Hazardous Substances in Electronic Equipment Directive(RoHS II)
